 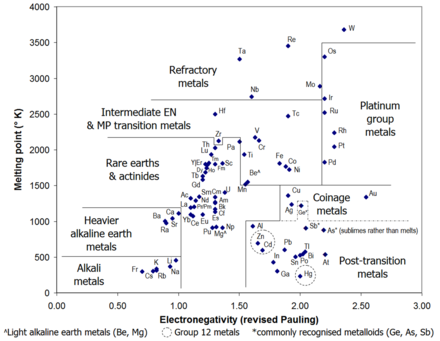  
The purpose of this chapter is to review the thermodynamic concepts pertinent to gas–metal reactions and then to describe the construction of the thermodynamic diagrams most often used in corrosion research, and to present illustrative examples of their application. The complexity of the corrosion phenomena usually dictates that the thermodynamic analysis be represented in graphical form. An important tool in the analysis of such problems is, of course, equilibrium thermodynamics which, although not predictive, allows one to ascertain which reaction products are possible, whether or not significant evaporation or condensation of a given species is possible, the conditions under which a given reaction product can react with a condensed deposit, etc. The situation is often complicated by the presence of liquid or solid deposits, which form either by condensation from the vapour or impaction of particulate matter. In practice, the corrosion problems to be solved are often complex, involving the reaction of multicomponent alloys with gases containing two or more reactive components. A sound understanding of high-temperature corrosion reactions requires the determination of whether or not a given component in a metal or alloy can react with a given component from the gas phase or another condensed phase, and to rationalize observed products of the reactions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
